|
When reading about smog production or ozone depletion, consider the bond strengths of the chemicals involved in this environmental problems. For example, the triple bond found in carbon monoxide is harder to break than one of the double bonds found in carbon dioxide. For this reason, it is more difficult to break or cleave a multiple bond than it would be a single bond or a bond of a lower order (when comparing the same connected atoms).  
Density is defined as the mass per unit volume. Typical densities of various substances are at atmospheric pressure. Density of Antimony Density of Antimony is 6.697g/cm3. Multiple bonds involve more that one type of connection. Electron configuration of Antimony is Kr 4d10 5s2 5p3. If an octet can be achieved with a single bond, do not incorporate multiple bonds into the structure. If a small amount of phosphorus is added to a pure silicon crystal, one of the valence electrons of phosphorus becomes free to move around (free electron) as a surplus electron. Its atomic symbol comes from its Roman name: stibium. Silicon of Group IV has four valence electrons and phosphorus of Group V has five valence electrons. Antimony has an electron configuration of Kr4d105s25p3. Antimony (Sb) was probably the first of the pnicogens to be obtained in elemental form and recognized as an element. Let's look at a few molecules that utilize multiple bonds:Ītoms that could employ multiple bonds would be oxygen, nitrogen, sulfur, phosphorus, and carbon. It lies below arsenic, and above bismuth, and it has properties similar to both of those elements. C) How many valence electrons does antimony have D) Underline the valance electrons in the shorthand Chapter 10 question 43 foundations of college chemistry 15e Look for antimony in the periodic table. B) Write the shorthand electron configuration for an atom of antimony. Although it is mathematically feasible, halogens will not use multiple bonds to become stable either. A) write the complete electron configuration for an atom of antimony. Fragments of a Chaldean vase made of antimony have been estimated to date to about 4000 bce. Electrons per Energy Level: 2,8,18,18,5 Shell Model Ionic Radius: 0. History The ancients were familiar with antimony both as a metal and in its sulfide form. It occurs chiefly as the gray sulfide mineral stibnite (Sb 2 S 3 ). Please note the rules above on which atoms will never achieve octets (hydrogen and boron). Antimony is a lustrous silvery bluish white solid that is very brittle and has a flaky texture. Antimony reacts with oxygen to give a trivalent oxide Sb2O3, a pentavalent. If a single bond does not work, then you may incorporate a multiple bond. The electron configuration of antimony is Kr4d105s25p3, with 5 valence electrons. silicon (Si), and germanium (Ge), antimony (Sb), arsenic (As). 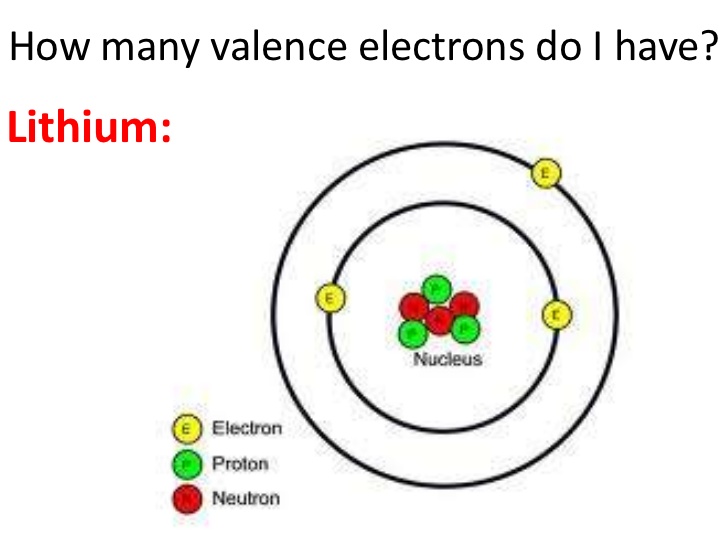
In covalent bonding, using double (four total electrons) or triple (six total electrons) is an option in order to achieve a stable octet. Valence electrons contribute to chemical reactions and bonding within the structure of a.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
